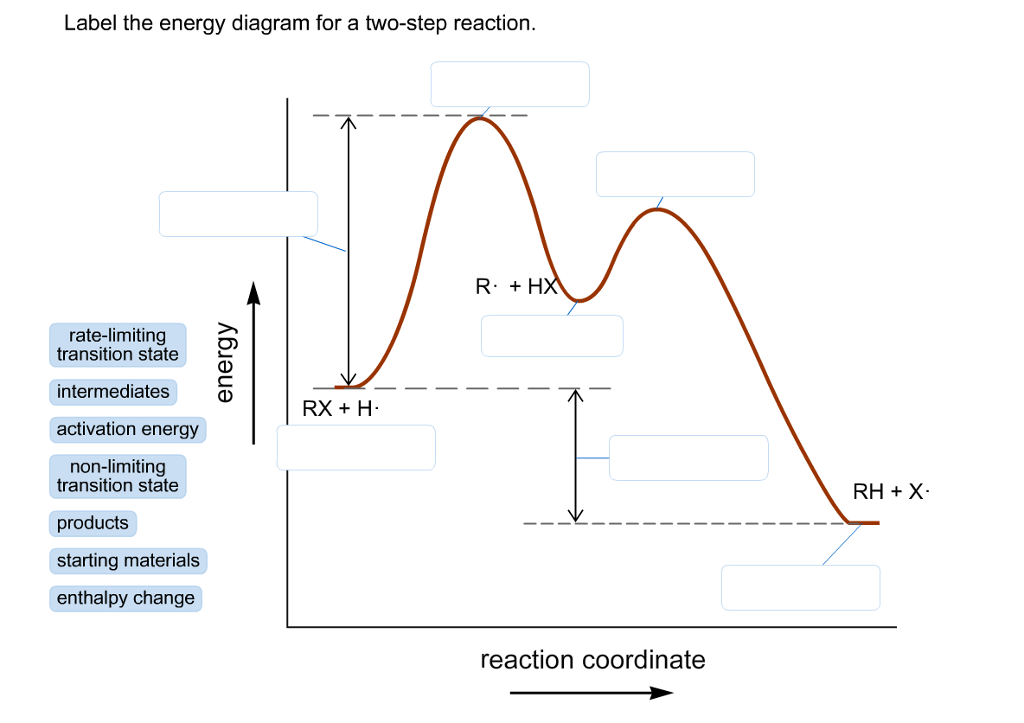
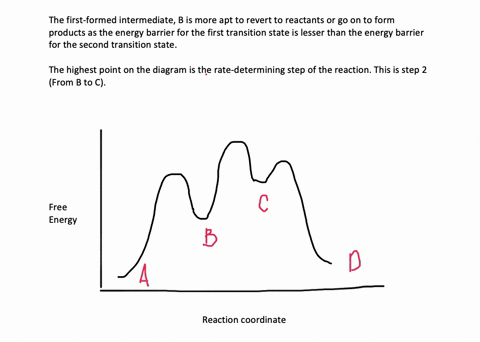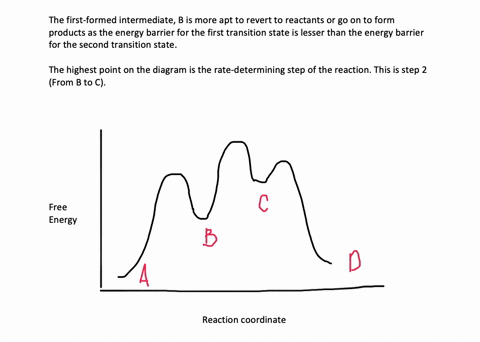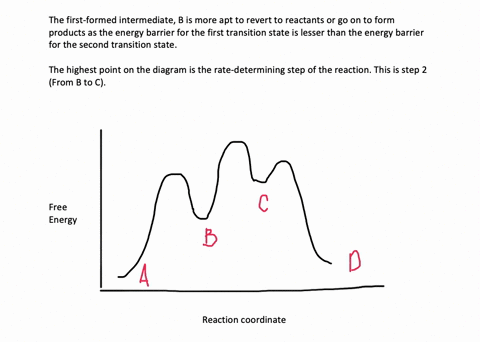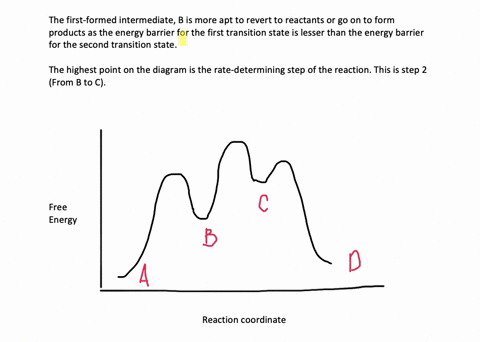
35 label the energy diagram for a two-step reaction.
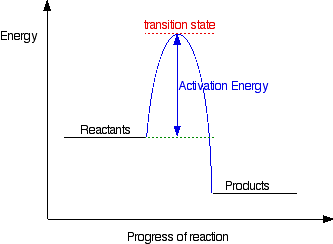
We're being asked to label the given energy diagram.. Recall that an energy diagram is usually read from left to right.The components of a two-step energy diagram are: • Reactants: are placed on the left/beginning of the energy diagram • Products: are placed on the right/end of the energy diagram • Non-limiting transition state: is the transition state with the lowest energy in the ... Draw the enthalpy diagram for the reaction and classify exothermic or endothermic. 2F_2 (g) + 2H_2O (l) to 4HF (g) + O_2 (g) An endothermic reaction is found to have an activation energy of 100 kJ.
Question: Label the following reaction energy diagram for a catalyzed and an uncatalyzed process. Transition State Transition State Transition intermediate Reaction Intermediate Transition State Transition Uncatalyzed Transition State Reactants Products Catalyzed Ea (fwd) no catalyst Uncatalyzed AHxn < 0 Potential energy Ea (rev) no catalyst Ea ...

Label the energy diagram for a two-step reaction.
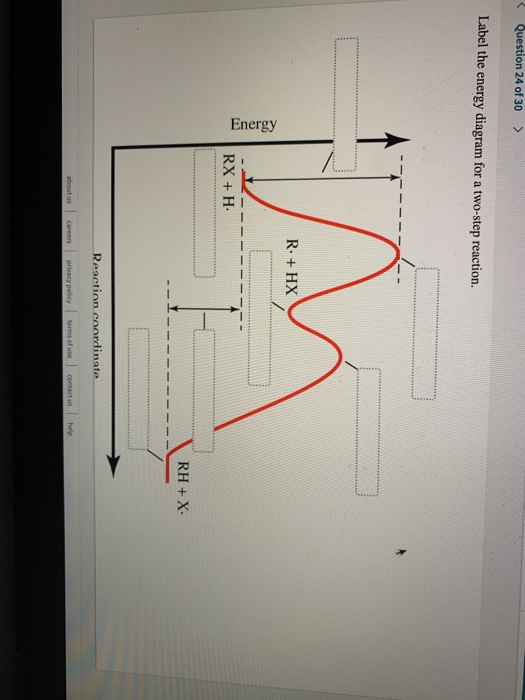
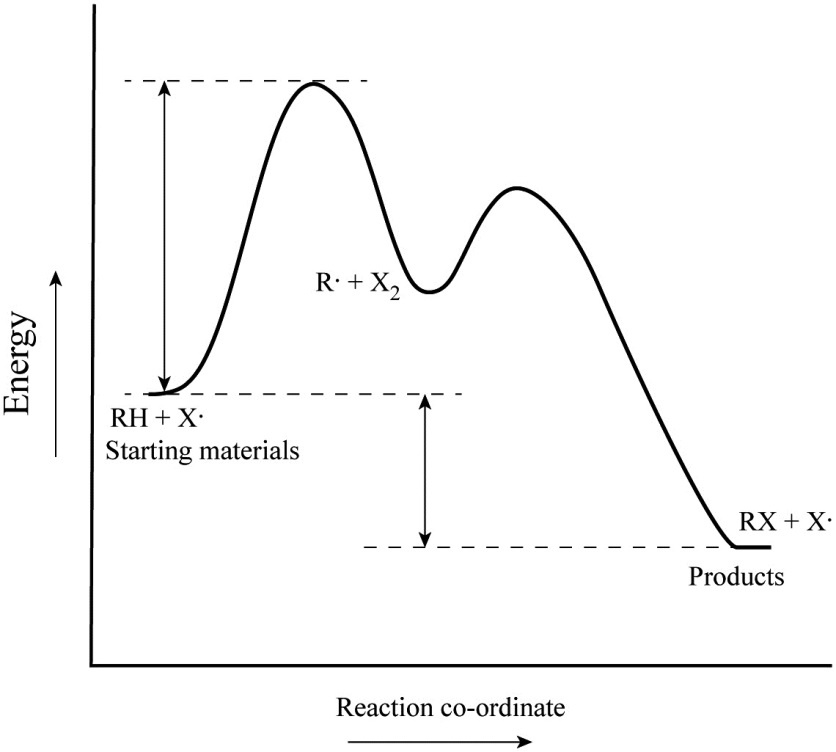
65AP. Draw a reaction energy diagram for a two-step reaction that has an endothermic first step and an exothermic second step. Label the reactants, transition states, reaction intermediate, activation energies, and enthalpy differences. Step-by-step solution. 100% (4 ratings) for this solution. Chapter 3, Problem 18P is solved. View this answer. The reaction of 2-methylpropene (or isobutylene) with HBr, as depicted above, is really a 2-step process. Draw this mechanism again and in each of the two steps label both the nucleophile and the electrophile (so, that's four labels). Problem EA1.2. Draw a reaction progress diagram for the reaction of 2-methylpropene with hydrogen bromide. Draw an energy diagram for a reaction that has 2 steps where the second step is the rate limiting step and is overall exothermic (label all parts of; Question: 2. Draw an energy diagram for a reaction that is one step, is endothermic, and has a very large activation energy (label all parts of the diagram).
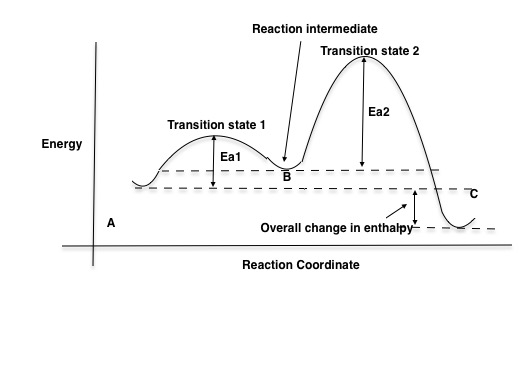
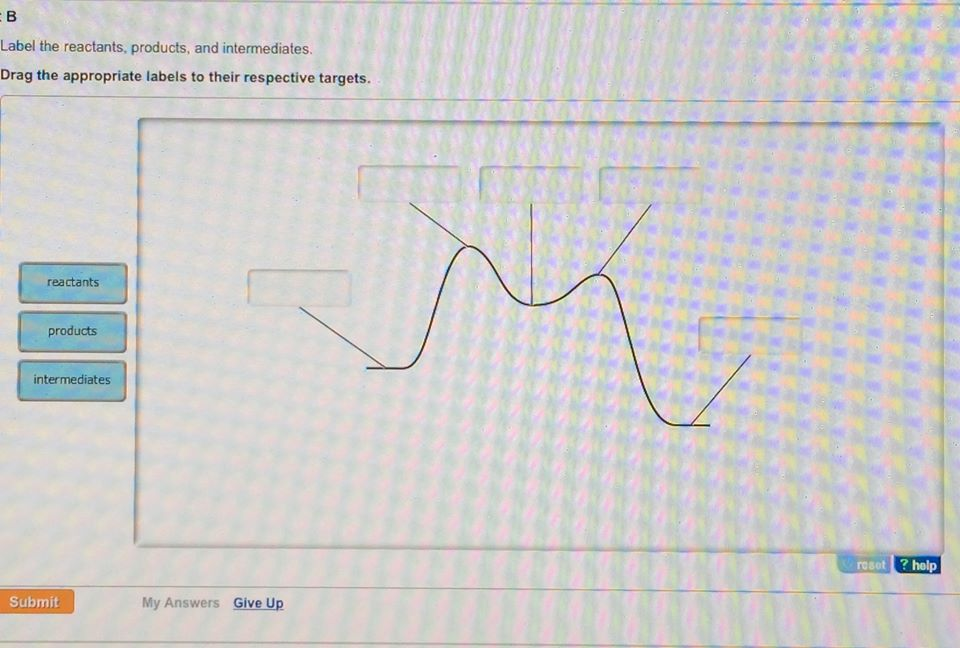
Label the energy diagram for a two-step reaction.. The reaction whose potential energy diagram is shown in the figure is a two-step reaction. The activation energy for each step is labeled E a1 and E a2 .Each elementary step has its own activated complex, labeled AC 1 and AC 2 .Note that the overall enthalpy change of the reaction is unaffected by the individual steps, since it depends only on the initial and final states. a) Label the electrophile and the nucleophile. b) Does the reaction proceed via S N 1 or S N 2 mechanism? c) Draw the products and complete mechanism of the reaction and identify the rate determining step. d) Is there an intermediate in this reaction? If yes, show its structure and position on the energy diagram. The following is an energy diagram for the conversion of A → B → C. The energies of activation and ∆H's for each step are also given. Calculate ∆H overall as shown on the energy diagram for A → B → C. 1. Identify the general shape of the energy diagram Energy should conserve for any chemical reaction. The reaction in question is exothermic (releases heat) hence its products shall have chemical potential energies lower than that of its reactants- some of the potential energies have been converted to thermal energy during the reaction process.
Draw a potential energy (E p ) diagram for a reaction in which ∆H = 80 kJ/mol and E a = +28kJ/mol. Label the axes, activation energy, ∆H, site of the activated complex, reactants and products. In the term S N 2, S stands for 'substitution', the subscript N stands for 'nucleophilic', and the number 2 refers to the fact that this is a bimolecular reaction: the overall rate depends on a step in which two separate molecules (the nucleophile and the electrophile) collide. A potential energy diagram for this reaction shows the ... 5. Question 5 - The Energy Diagram of SN2 reaction: Draw an energy diagram for the following S N 2 reaction. Label the axes, the Ea, the ΔH° and the transition state of the reaction.Assume the reaction is exothermic and ΔH° = -75 kJ/mol and Ea = 50 kJ/mol. Draw the structure of reactants and products on the diagram.You can put the reactants at any energy level and then draw the rest as ... This photo about: Label This Energy Diagram, entitled as Label The Energy Diagram For A Two Step Reaction - Label The Energy Label This Energy Diagram - also describes Label The Energy Diagram For A Two Step Reaction - Label The Energy and labeled as: ], with resolution 3102px x 1093px
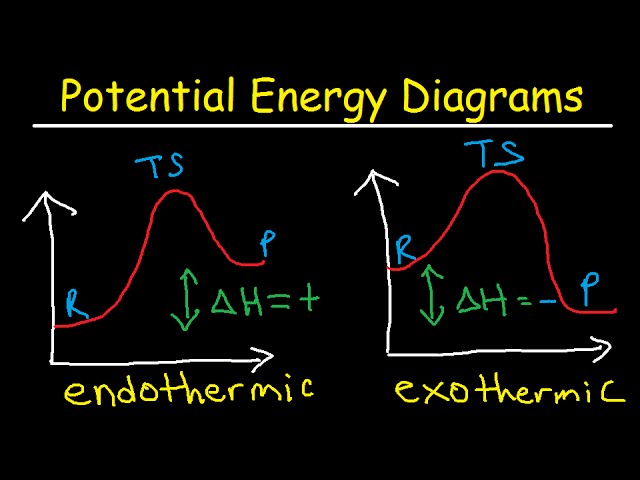
Label ΔH as positive or negative. Figure shows the energy level diagram for the reaction between methane and oxygen. Based on Figure, the following information can be obtained. (a) The reaction between methane and oxygen to form carbon dioxide and water is an exothermic reaction. (b) During the reaction, the temperature of the mixture increases. A Two-Step Reaction Mechanism We draw an energy diagram for each step, and then combine them in an energy diagram for the overall two step mechanism. Label the energy diagram (7 bins) and indicate which reaction corresponds to the energy diagram. Label the energy diagram (7 bins) and indicate which reaction corresponds to the energy diagram. Draw an energy diagram for a two-step reaction that is exothermic overall, and consists of a fast but endothermic first step, and a slow but exothermic second step. Indicate DG rxn , as well as DG 1 * and DG 2 * for the first and second activation energies, respectively. This is part 3 of a four part series in the Energy Diagram Module. Stay tuned for Part 4! Click on the following links to see earlier parts: Part 1. Part 2. Sometimes reactions are more complex than simply a transition state (Graph 3), which would represent a single step in the reaction mechanism.
1. Identify the major product for the reaction below and provide a detailed reaction mechanism to account for its formation: Label the rate determining step (RDS)* HCI- Major Product Using the reaction above, fill in the reaction energy diagram and clearly indicate and CLEARLY label each of the following: 2.
here for the reaction. Coordinate diagram. Put two step verification in which the first step, Andrea Janek, the second step is extra, Janek. And the order reaction is and a sonic labels are re intense. Products, intermediates and chronic transition is start stairs. So this is the reaction co ordination diagram in which this is for free energy, the bikes and X axis for the the reaction coordinates.
Below is an energy diagram illustrating the difference in a catalyzed reaction versus an uncatalyzed reaction. Label the energy diagram and answer the question that follows% (1). Catalyzed reactions have a lower activation energy (rate-limiting free energy of activation) than the corresponding uncatalyzed reaction, resulting in a higher ...
Consider the one-step conversion of F to G. Given that the reaction is endothermic by 5 kcal/mol and that the energy difference between G and the transition state for the process is 15 kcal/mol, sketch a reaction-energy diagram for this reaction. Make sure to show how the given energy differences are consistent with your sketch.

Welcome To Modern Organic Chemistry Chapter 4 The Study Of Chemical Reactions Organic Chemistry 5 Th Edition L G Wade Jr Ppt Download
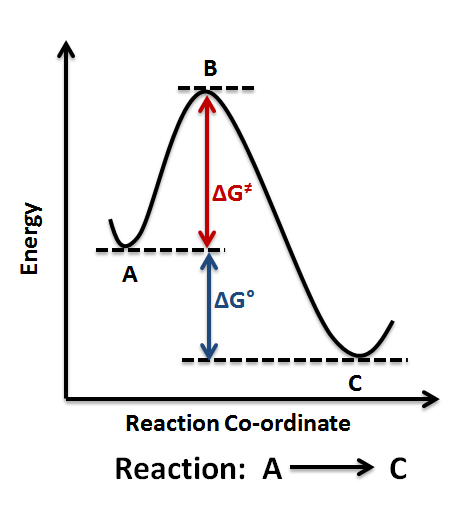
Sketch an energy diagram for a two-step reaction in which both steps are exergonic and in which the second step has a higher-energy transition state than the first. Label the parts of the diagram corresponding to reactant, product, intermediate, overall ΔG ‡, and overall ΔG°.
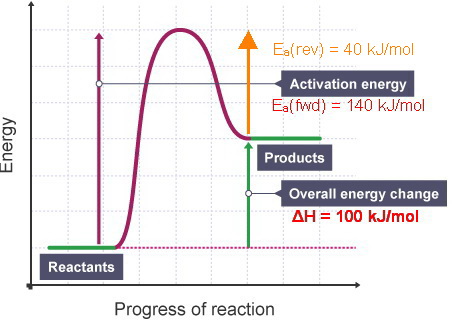
A Reaction Is Endothermic With H 100 Kj Mol If The Activation Enthalpy Of The Forward Reaction Is 140 Kj Mol What Is The Activation Enthalpy Of The Reverse Reaction I Know The Answer Is
57CP. Draw an energy diagram for each reaction. Label the axes, the starting material, product, transition state, ΔH°, and E a. a. A concerted, exothermic reaction with a low energy of activation. b. A one-step endothermic reaction with a high energy of activation. c. A two-step reaction.
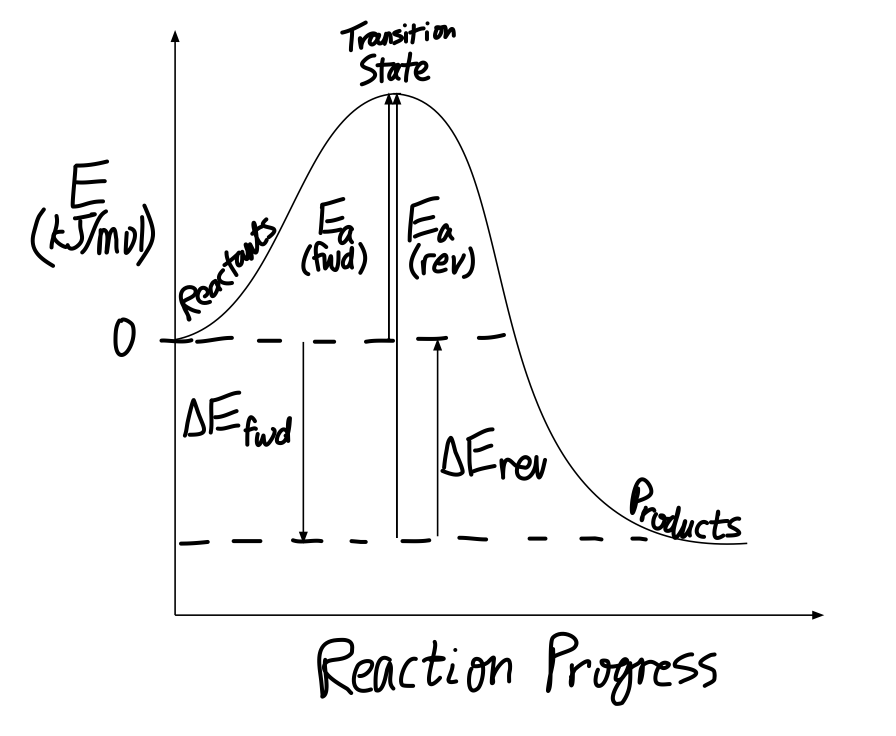
How Would You Draw And Label Energy Diagrams That Depict The Following Reactions And Determine All Remaining Values Place The Reactants At Energy Level Zero Socratic
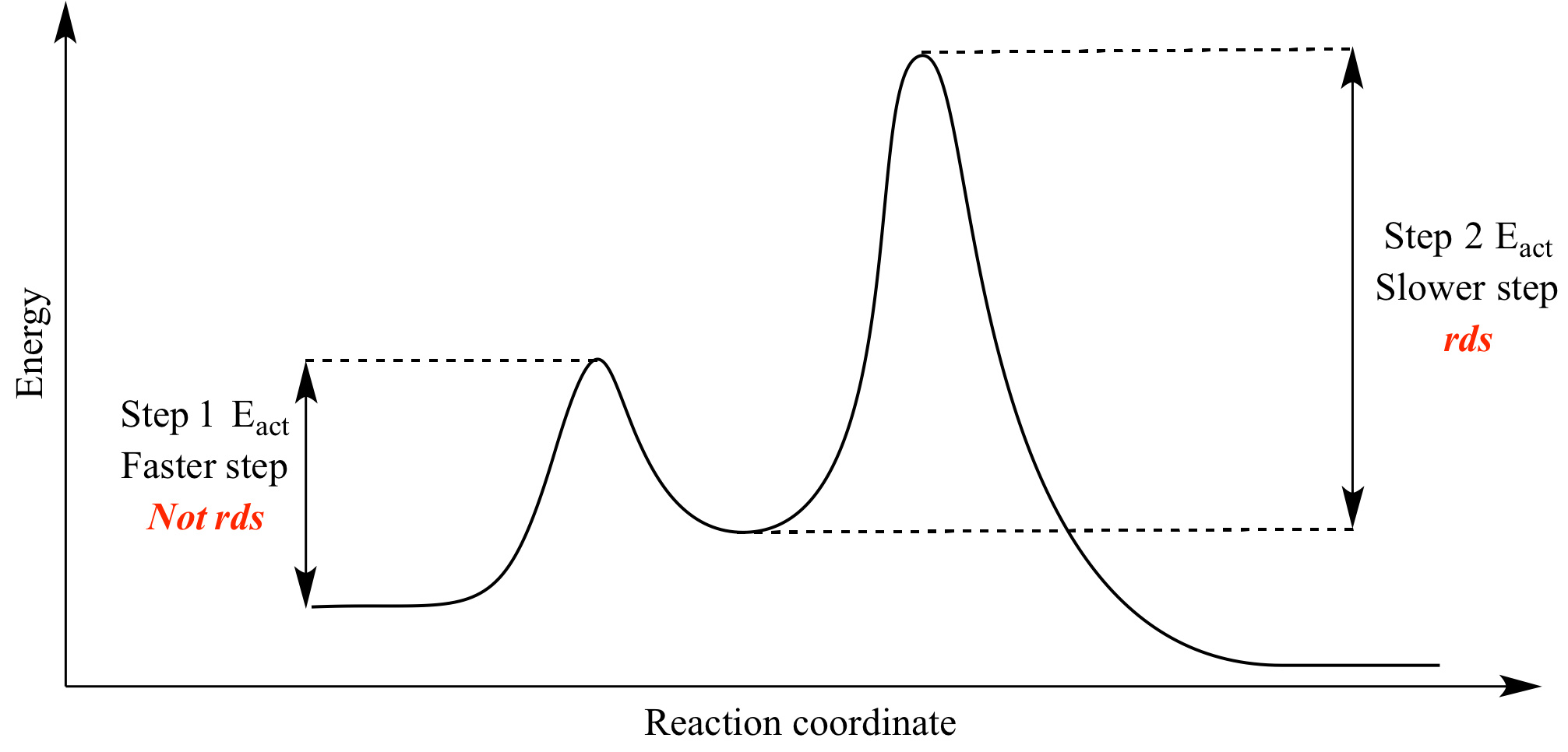
is the complex created in the first reaction, while is the activated complex created in the second reaction. Thus, for this two-step process, there are two activated complexes. Example: Draw the potential energy diagram for the following multi-step reaction . Properly label the diagram. Solution: Rate of Reaction is Determined by Slowest Step
Draw an energy diagram for a reaction that has 2 steps where the second step is the rate limiting step and is overall exothermic (label all parts of; Question: 2. Draw an energy diagram for a reaction that is one step, is endothermic, and has a very large activation energy (label all parts of the diagram).
The reaction of 2-methylpropene (or isobutylene) with HBr, as depicted above, is really a 2-step process. Draw this mechanism again and in each of the two steps label both the nucleophile and the electrophile (so, that's four labels). Problem EA1.2. Draw a reaction progress diagram for the reaction of 2-methylpropene with hydrogen bromide.
65AP. Draw a reaction energy diagram for a two-step reaction that has an endothermic first step and an exothermic second step. Label the reactants, transition states, reaction intermediate, activation energies, and enthalpy differences. Step-by-step solution. 100% (4 ratings) for this solution. Chapter 3, Problem 18P is solved. View this answer.
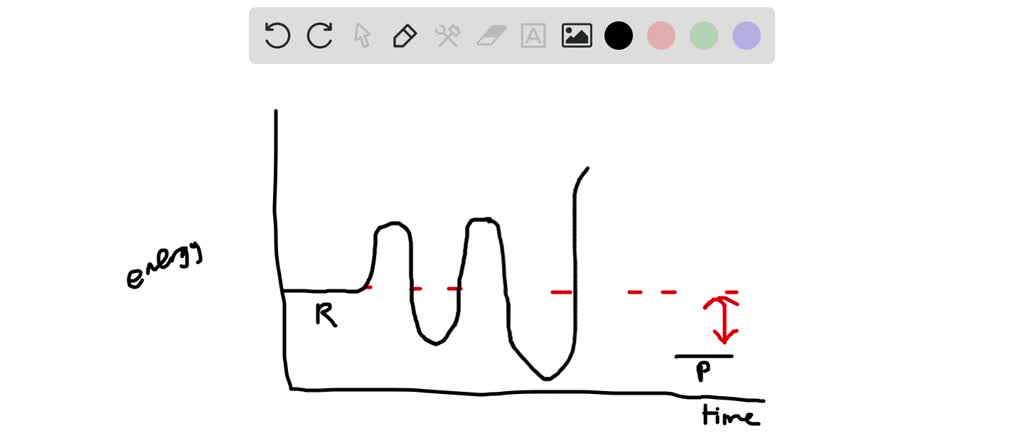
Solved On Your Paper Draw An Energy Diagram For A Two Step Endothermic Reaction Label All Of The Following Areas On The Graph For The Forward Reaction Reactants Products Activation Energy Energies Transition State S Intermediate S
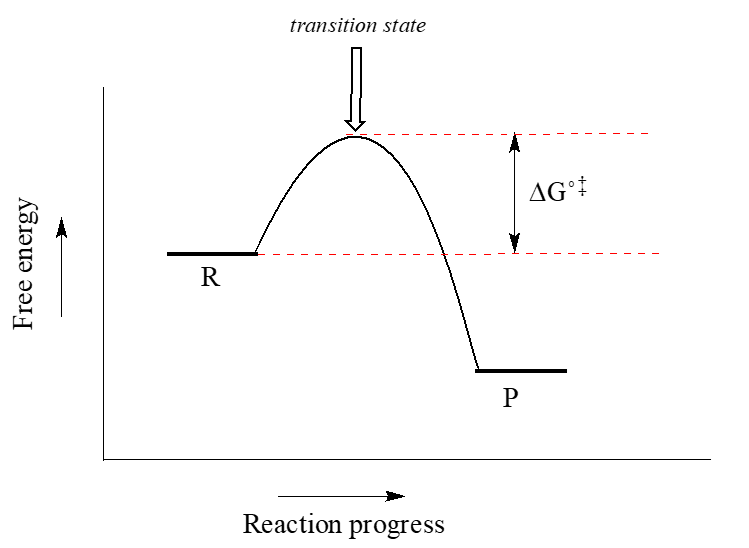















0 Response to "35 label the energy diagram for a two-step reaction."
Post a Comment